
Safety Audit Study – Abbott Healthcare Limited.
Project Number: 24-738.02.165-ahpl-jgi-saau
Safety Audit Serial No: 165
- Project Overview
A Safety Audit was carried out to systematically evaluate the effectiveness of the Occupational Safety and Health (OS&H) management system and to identify potential hazards, unsafe conditions, and non-compliances associated with the operations at Abbott Healthcare Limited, Jhagadia, Gujarat. The audit focused on assessing risks to personnel, plant, equipment, and operations, with emphasis on fire, explosion, chemical, and occupational health hazards arising from storage, transfer, and associated activities.
The objective of the Safety Audit was to critically appraise existing safety practices, engineering and administrative controls, emergency preparedness, and statutory compliance, and to verify their alignment with applicable legal and corporate safety requirements. The audit aimed to identify gaps, recommend risk-based corrective actions, and support continual improvement in safety performance, thereby minimizing the likelihood and consequences of accidents, fires, and explosions. The study was conducted in accordance with IS 14489:2018 – Code of Practice on Occupational Safety and Health Audit.
The Safety Audit involved a comprehensive review of critical documents and on-site practices, including safety policies, SOPs, training records, statutory licenses, inspection and maintenance records, emergency plans, previous audit findings, and legal compliance registers. The study also included site walkthroughs, interviews with operating personnel, evaluation of emergency response arrangements, and risk-based prioritization of recommendations to enhance personnel safety, regulatory compliance, and safe, reliable operation of the facility without adversely impacting operational efficiency.
- Objective
The objective of the Safety Audit was to:
- To carry out a systematic, critical appraisal of all potential hazards involving personnel, plant, services and operation method; and
- To ensure that OS&H system fully satisfy the legal requirements and those of the company’s written safety policies, objectives and progress.
- Methodology
- Safety Audit Concept
In all those industrial units, where a safety program is oriented towards controlling work injuries, obviously, the tool used for measuring company safety performance will be the frequency and severity of work injuries, as compared to employee exposure. Apart from being restrictive, this method of evaluating company safety efforts is reactive, and not proactive. To correct this deficiency, modern program evaluation methods take into consideration all consequences of undesirable events.
The most commonly used modern tool for program evaluation is the “Safety-Audit”, which is now a statutory requirement for units dealing with hazardous materials and process. While carrying out safety audit, an attempt is made to identify and assess management efforts towards each element of the safety Programme, evaluate this against standards established by the company, national, international and statutory requirements. In the process, deficiencies are identified and corrective measures undertaken. Periodically, follow-up audits help to monitor implementation of safety audit recommendations. Thus it can be seen that Safety Audit is a complete, result oriented proactive tool to evaluate company safety efforts.
- Safety Audit Methodology
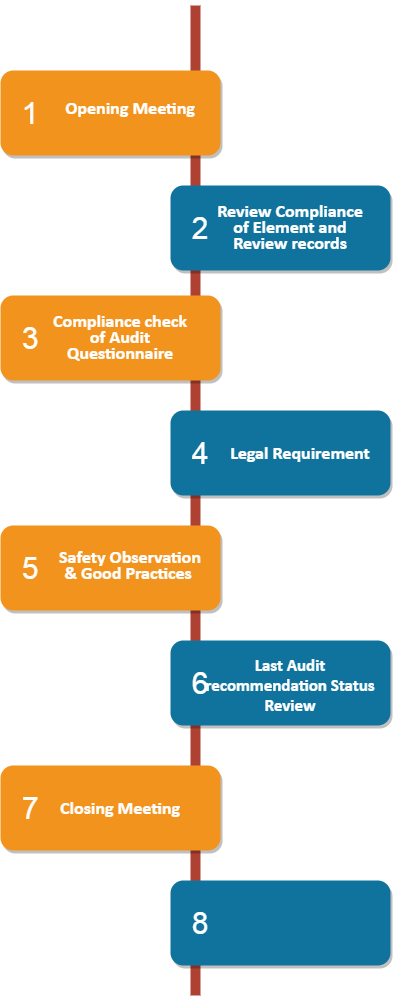
The overall methodology followed during audit is as below:
- Opening Meeting
- The audit scope, objective, methodology and audit schedule was discussed by auditors with auditee during opening meeting.
- The manufacturing process and existing OS & H system was explained by auditee to auditors
- Examination
- Compliance review of all 76 elements of occupational safety and health system (OS & H) of IS-14489. Wherever applicable, Procedure/ Guideline/ Document, Training matrix, MIS, Compliance Audit is reviewed of each Element.
- 34 nos. of Records review as per IS:14489
- Compliance check of Audit Questionnaire
- Review License as per applicable Legal Citation
- Review the opening/ closing status of Last Audit recommendation
- Site Round
- Site round in various department for observing any lagging and good practices implemented by Organization
- Closing meeting
- Closing meeting was carried out after completion of the audit
- The audit observations, recommendations and elements compliance report was discussed in the meeting
Key distinctive characteristics of Safety Audit:
- The audit is divided into 7 Segments, as below:
- Elements of OS&H – Evaluation of the overall Occupational Safety & Health management system, including policy, organization, procedures, training, and safety culture.
- Records Examination – Verification of safety-related records such as training logs, inspection reports, permits, maintenance documents, and incident records to confirm implementation of safety systems.
- Audit Questionnaire – Structured assessment using standard checklists to evaluate compliance with IS 14489 requirements and effectiveness of safety practices.
- Legal Requirement – Review of statutory compliances under applicable safety, health, and hazardous chemicals regulations to ensure adherence to legal obligations.
- Good Practices – Identification and documentation of effective safety practices that exceed statutory or standard requirements and contribute to improved safety performance.
- Site Round Observation – Physical inspection of plant areas to identify unsafe conditions, unsafe acts, and potential hazards during actual operations.
- Last Audit Status Review – Assessment of the implementation status and effectiveness of corrective actions from previous safety audit recommendations.
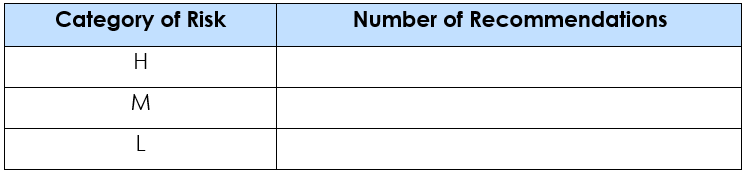
- Audit Recommendations are provided in the following format ensuring completeness and categorization.

- A comprehensive compliance summary is provided in the following format.
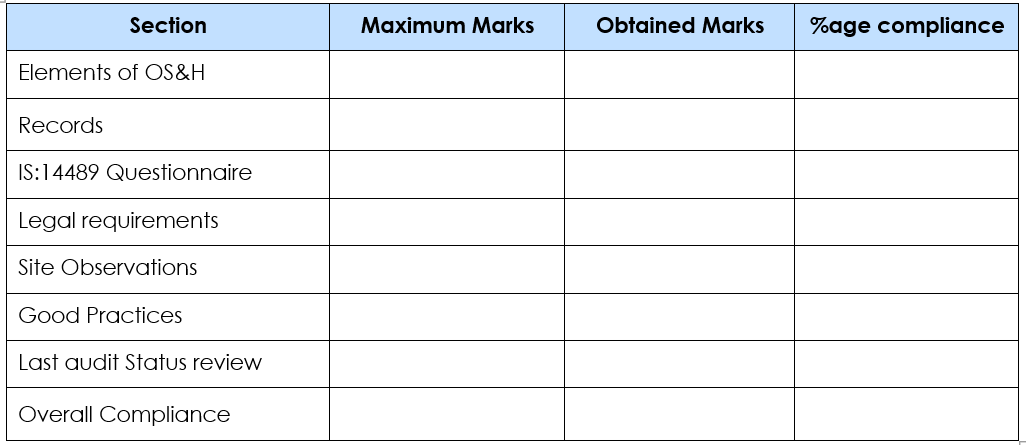
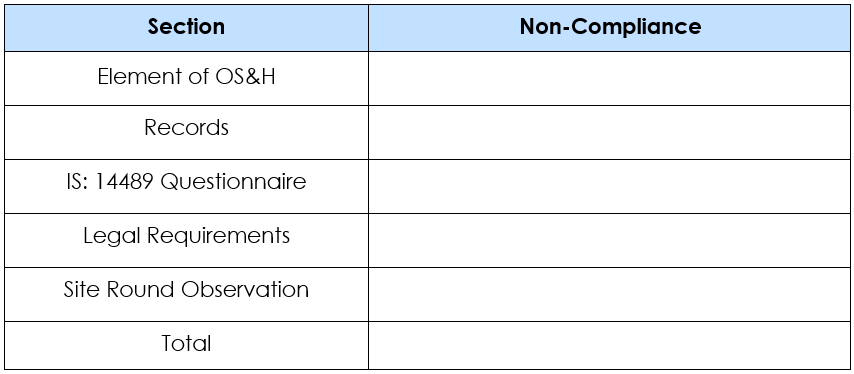
- In the Safety Audit, not only the non-compliances were identified but also, the good practices that the company is following were also highlighted showing the dedication of the Abbott Healthcare Limited to ensure robust occupational health and safety.
- Key Outcomes
- The overall compliance of Abbott Healthcare Limited is well above 95% showcasing robust OS&H management. Hence, The OS&H management system of Abbott Healthcare is rated in “Excellent” category. The OS&H management system at Abbott healthcare Limited is found able to march towards improvements.

- Comprehensive OS&H Performance Evaluation – The audit provided a structured assessment of the Occupational Safety & Health management system, highlighting strengths, gaps, and areas for improvement in line with IS 14489 requirements.
- Almost 30% of the recommendations were hardware related improvements while 70% of the recommendations were administrative changes/improvements.
- Identification of Compliance Gaps and Risks – Statutory non-compliances, unsafe conditions, and procedural gaps were identified through document review and site observations, with risks categorized as High, Medium, and Low.
- Actionable, Risk-Based Recommendations – Practical recommendations were developed and prioritized to mitigate identified risks and enhance safety, reliability, and emergency preparedness of operations.
- Conclusion
The Safety Audit concluded that the Occupational Safety and Health (OS&H) management systems and safety practices at the facility are generally effective in controlling risks associated with the storage and handling of hazardous materials and related operations. The audit provided a structured evaluation of statutory compliance, operational controls, and emergency preparedness, and identified specific gaps requiring improvement through risk-based recommendations. Overall, the study demonstrates the importance of periodic safety audits, systematic implementation of corrective actions, and adherence to applicable legal and standard requirements in ensuring safe, reliable, and regulatory-compliant plant operations.



